Změny v mozkovém objemu při monokulární slepotě s pozdním nástupem – studie volumetrického zobrazení
Změny v mozkovém objemu při monokulární slepotě s pozdním nástupem – studie volumetrického zobrazení
Cíl: Narůstá počet studií, ve kterých bylo prokázáno, že lidé, kteří jsou od narození slepí, mají pravděpodobně určité změny v mozku, ačkoli neuroanatomické a funkční dopady monokulární slepoty s pozdním nástupem (late-onset monocular blindness; LMB) nejsou dobře známy. Cílem naší studie bylo měření objemů mozkových struktur u jedinců s LMB a jejich srovnání s hodnotami u zdravých jedinců s cílem prozkoumat vlastní neuroadaptivní mechanizmy. Materiál a metodika: Byla hodnocena skupina 13 pacientů s LMB po úrazu jednoho oka a kontrolní skupina 13 subjektů, kteří věkem a pohlavím odpovídali pacientům s LMB. Všichni účastníci studie podstoupili kompletní oftalmologické vyšetření. Pomocí 1,5T zařízení byla u všech subjektů provedena MR mozku a volumetrická data byla analyzována pomocí postupů MRIvolBrain a MRICloud. Výsledky: V porovnání se zdravými kontrolami jedinci s LMB vykazovali významně vyšší objem nucleus accumbens a nižší objem středního okcipitálního gyru a cuneu (Cu). Statistická analýza slepých očí a očí se zrakem u jedinců s LMB ukázala nízký poměr cornu ammonis k intrakraniální dutině a nízký objem Cu na straně slepých očí. Při srovnání mozkových objemů na straně očí se zrakem u lidí s LMB a ipsilaterálních mozkových objemů u kontrol jsme zjistili nižší objem supramarginálního gyru a vyšší objem subicula ve skupině LMB než v kontrolní skupině. Závěr: LMB pravděpodobně vede k strukturním změnám v určitých oblastech mozku, např. v limbickém systému a zrakovém kortexu, což potenciálně hraje roli v neuroadaptivních mechanizmech.
Klíčová slova:
pozdní nástup – monokulární slepota – zobrazení mozkových struktur – mozkové objemy
Authors:
E. Ö. Öktem 1; F. Aslan 2; O. Ozen 3
Authors‘ workplace:
Department of Neurology, Alanya Alaaddin Keykubat University, Antalya, Turkey
1; Department of Opthalmology, Alanya Alaaddin Keykubat University, Antalya, Turkey
2; Department of Radiology, Alanya Alaaddin Keykubat University, Antalya, Turkey
3
Published in:
Cesk Slov Neurol N 2023; 86(2): 121-127
Category:
Original Paper
doi:
https://doi.org/10.48095/cccsnn2023121
Overview
Aim: An increasing number of studies have demonstrated that persons who are blind from birth may have some alterations in the brain, although the neuroanatomical and functional effects of late-onset monocular blindness (LMB) are not well known. In our study, we aimed to measure the volumes of cerebral structures in LMB individuals and compare with healthy subjects, in order to investigate the underlying neuro-adaptive mechanisms. Material and methods: A study group with 13 LMB patients due to an isolated ocular trauma and a control group with the 13 age- and sex-matched healthy individuals were evaluated. All participants underwent a full ophthalmological examination. Brain MRI examination of the participants was performed using a 1.5-T MRI device and volumetric data were analyzed through the MRIvolBrain and MRICloud procedures. Results: Compared with healthy controls, individuals with LMB showed significantly increased accumbens volumes and decreased volumes of the middle occipital gyrus and cuneus (Cu). In the statistical analysis between the blind eyes and sighted eyes of LMB individuals, the ratio of cornu ammonis to the intracranial cavity and Cu volumes were low on the blind side. In a comparison of cerebral volumes on the sighted side of the LMB group and ipsilateral cerebral volumes of controls, we found lower supramarginal gyrus volumes, higher subiculum volumes in the LMB group than in the control group. Conclusion: LMB may lead to structural alterations in specific brain regions, such as the limbic system and visual cortex, which play a potential role in neuro-adaptive mechanisms.
Keywords:
late-onset – monocular blindness – brain structural imaging – cerebral volumes
Introduction
Blindness, also known as the inability to respond to light inputs from the environment, not only limits a person’s quality of life but also poses a threat to public health on a global scale. Generally, blindness can be categorized into early-onset and late-onset blindness, as well as monocular and binocular blindness. Ocular trauma, cataracts, glaucoma, age-related macular degeneration (AMD), keratoconus, amblyopia and optic atrophy, are some conditions that can lead to late-onset monocular blindness (LMB) [1]. According to statistics provided by the World Health Organization (WHO), blindness and other forms of visual impairment, impact at least 2.2 billion persons around the globe. There are preventable causes for one billion of these cases, which has led to the establishment of a global programme known as “Vision 2020 – The right to see”, which seeks to understand how to reduce preventable blindness [2–4]. Clinicians are presented with a significant obstacle when attempting to devise treatment plans for blindness, because the condition often results in irreversible damage. We believe that numerous treatments developed to treat blindness, such as retinal implants, gene therapy and applications involving stem cell research, can only be successful if the neuroanatomical and functional abnormalities that are associated with blindness, are well understood [5,6]. Blind persons may be able to make up for their impaired vision in some way by making better use of their remaining senses, such as hearing and touch [7,8]. In addition to this clinical compensation, some neurophysiological compensatory alterations may occur in the regions of the brain that are involved in somatosensory and auditory processing. In early-onset monocular individuals, it has been established that those other cerebral regions, including the hippocampus, in addition to somatosensorial and auditory regions, are impacted in comparison with sighted individuals [9]. In a morphometric investigation, it was shown that blind individuals have higher navigational skills that controls the route-learning, along with a considerable volume increase in the hippocampus. This was the case regardless of whether the blindness was congenital or acquired by the individual [10]. A recent functional MRI (fMRI) study showed that early-onset monocular individuals had signal modifications within the auditory cortex that were compatible with the evidence of more effective processing of the auditory input [11]. It is well known that early-onset blindness is associated with several neuroanatomical and neurofunctional modifications; on the other hand, the nature of these alterations in persons with late-onset advanced monovision is yet unknown [12,13]. We aimed to measure the cerebral structures of LMB individuals and compare them with healthy subjects, in order to investigate neuro-adaptive modifications and the underlying mechanisms in LMB.
Methods
Patient selection and study protocols
This prospective designed, case-control study was conducted at Alanya Alaaddin Keykubat University, Turkey between July 2020 and December 2020.
A study group with 13 monocular patients due to isolated ocular trauma and a control group with age- and sex-matched healthy individuals were evaluated. The study group consisted of patients who had been monocular for at least 5 years, with negative light perception in the blind eye.
Inclusion and exclusion criteria
Only participants between the ages of 20 and 75 years were included in the study. Healthy subjects with a refractive value between +1.5 and –1.5 dioptres, a best-corrected visual acuity of at least 20/20 and an axial lenght (AL) of \ 25 mm were included in the control group. Individuals with at least 5 years of monocular blindness, a refractive error between +1.5 D and –1.5 D in the sighted eye, a best-corrected visual acuity of least 20/20 and an AL \ 25 mm, were included in the study group.
Patients with concomitant neurological disorders that could influence cerebral volumetric measurement such as dementia, multiple sclerosis, Parkinson’s disease, stroke, high myopia, or retinal or macular pathology, a history of ocular surgery or trauma in the sighted eye, those with a history of glaucoma, optic neuropathy, uveitis, congenital blindness, blindness caused by other eye diseases (e. g., cataract, glaucoma, optic neuritis, macular degeneration, or ocular ischemic disease), those with a history of intracranial tumor, operation, substance or alcohol abuse, were excluded from the study.
Ophthalmological examination
All participants underwent a full ophthalmological examination, including best-corrected visual acuity (BCVA) measurement with the Snellen Scale, anterior segment biomicroscopic examination, intraocular pressure (IOP) measurement using Goldmann applanation tonometry, gonioscopy, fundus examination, central corneal thickness (CCT) (Nidek, Gamagori, Japan) measurement, visual field (VF) tests, AL measurement, spectral domain optical coherence tomography (SD-OCT) (Optovue Inc., Fremont, CA, USA), and retinal and choroidal evaluation. A TOPCON TRC 50 DX (Topcon Corporation, Tokyo, Japan) high-resolution digital retinal camera was used to assess the fundus. The participants also had an ophthalmic examination, including slit lamp biomicroscopy and ophthalmoscopy. Ophthalmologic examination data, including AL, anterior chamber depth, CCT and IOP were recorded in both groups.
Neuroimaging and volumetric measurement
Brain MRI examinations of the participants were performed using a 1.5-T MRI device (GE, SIGNA Explorer, General Electric, Milwaukee, WI, USA). Conventional brain MRI on three-dimensional fast spoiled gradient recalled acquisition in the steady state (3D-FSPGR) array plus T1-weighted 3D fast decaying gradient recall procedure, was applied for use in segmental-volume measurement. The parameters of the 3D T1 FSPGR array are TE 1.7 ms, TR 5.95 ms, flip angle 12°, acquisition matrix 256 × 256, field of view 256 mm2, number of slices 170, and slice thickness 1.0 mm.
MRICloud procedure
MR volumetric image data in the 3D T1 weighted image in the sagittal plane were downloaded and transferred to a personal computer. They were analyzed using the DTI Studio software to generate header (HDR) and image (IMG) analysis formats. HDR and IMG files of the analyzed patients were uploaded to a web-based module (BrainGPS) run online via the web interface. The MRICloud web-based module is an automated volumetric analysis system that provides reliable and consistent volumetric information of data [14,15]. We used the Adult_286labels_ 10atlases_V5L atlas to process our data and we uploaded the axial slice type of MRI data of all participants to the MRICloud database.
Volbrain procedure
Participants’ 3D T1 FSPGR sequence images downloaded in digital imaging and communication in medicine (DICOM) format, were converted to neuroimaging information technology initiative (NIfTI) file format. The file in NIfTI format was uploaded to the vol2Brain 1.0 database via a web browser and segmental volumetric calculation was performed. The atlas used for processing our data in the MRICloud was adult_286labels_10atlases_V5L.
The volBrain CERES database uses a fully automated segmentation technique in which the algorithm is based on multi-atlas-based tag merge segmentation technology. It gives the volume of subcortical brain structures in cm3 and calculates the percentage ratio of all structures to the whole brain [16,17].
In volBrain, in addition to brain white matter (WM), grey matter (GM), cerebrospinal fluid and lateral ventricles, subcortical structures such as basal ganglia including caudate nucleus, putamen, globus pallidus, nucleus accumbens, thalamus, amygdala, hippocampus (cornu ammonis [CA1-4] dentate gyrus [DG]), corpus callosum and subiculum (Sb) were calculated. In addition, volume/percentage ratio in the intracranial cavity (IC) and IC volumes were calculated. The volumes of cortical structures such as supramarginal gyrus (SMG) and occipital gyrus (OG) were measured. VolBrain gives the volume of subcortical brain structures in cm3 and calculates the percentage ratio of all structures to the whole brain.
In MRICloud subcortical white matter (OWM), cuneus (Cu), lingual gyrus (LG) volumes were also evaluated. The accuracy of registration and segmentations was visually checked. The findings were then statistically compared between the two groups. The sample images of volBrain and MRICloud segmentation are shown in Fig. 1, 2.
1 – putamen; 2 – thalamus; 3 – globus pallidus; 4 – hypothalamus; 5 – frontal horn of the lateral ventricle; 6 – amygdala; 7 – caudate nucleus
Obr. 1. VolBrain segmentace – (a) intrakraniální dutina; (b) klasifikace tkání (šedá a bílá hmota); (c) makrostruktury (mozkový kmen, levá
a pravá hemisféra a mozeček); (d) subkortikální struktury.
1 – putamen; 2 – talamus; 3 – globus pallidus; 4 – hypotalamus; 5 – frontální roh postranní komory; 6 – amygdala; 7 – nucleus caudatus

Obr. 2. 3D zobrazení cuneu v MRICloud.

Statistical analysis
In the study, descriptive data are shown as numbers and percentages in categorical data, and median and (minimum-maximum) values or mean ± standard deviation values in continuous data. The Fisher test was used to compare the categorical data. Measurement data were tested with the Kolmogorov-Smirnov tests for the assumption of normal distribution. The T-test in independent groups was used to compare normally distributed measurement data, and the Mann–Whitney U test was used to compare non-normally distributed measurement data. Since we considered the LMB pathophysiology and related volumetric differences as independent variables (to eliminate possible confounding effects related to the dependency of groups) although it is apparent in the same individual, we opted to use independent samples tests, such as the Mann Whitney U Test, instead of paired tests. P < 0.05 was accepted for statistical significance in all analyses, which were performed using the SPSS programme version 20 (IBM, Armonk, NY, USA).
Results
In this study, 13 (10 male) LMB patients in the study group and 13 (10 male) individuals in the control group were examined. The mean age of the study group was 39 years, similar to the mean age of the control group at 40, and there was no statistically significant difference between the groups in terms of age and sex (P > 0.05) (Tab. 1). In the study group, the shortest duration of LMB was 8 years and the maximum duration was 32 years, with an average of 20.8 years. All subjects in the study were right-handed.
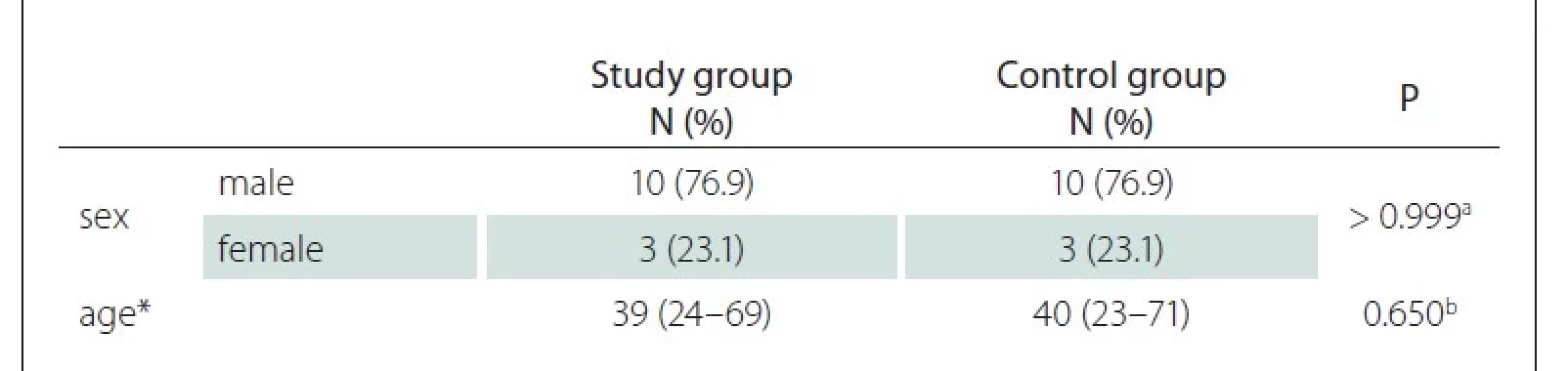
N – number
In the statistical analysis between the blind eyes and sighted eyes of LMB individuals, the ratio of hippocampus cornu ammonis 2–3 (CA2-CA3) to the IC and Cu volumes were low in the blind side (P < 0.05) (Tab. 2). There was no significant difference in other parameters (P > 0.05) between both groups of LMB patients.
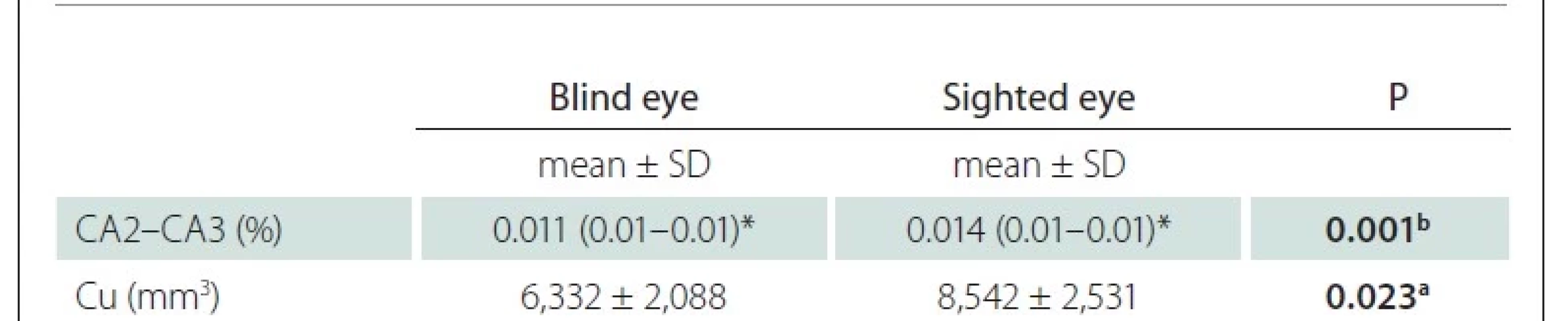
Cu – cuneus; LMB – late-onset monocular blindness; SD – standard deviation
We also compared the blind eyes of LMB individuals with the age- and sex-matched ipsilateral data in the healthy control group. The accumbens (Acc) volume and its ratio to the IC were found to be higher in the LMB group, and the volumes of the middle occipital gyrus (MOG) and Cu were found to be lower than those in the control group (Tab. 3). There was no significant difference in other parameters.
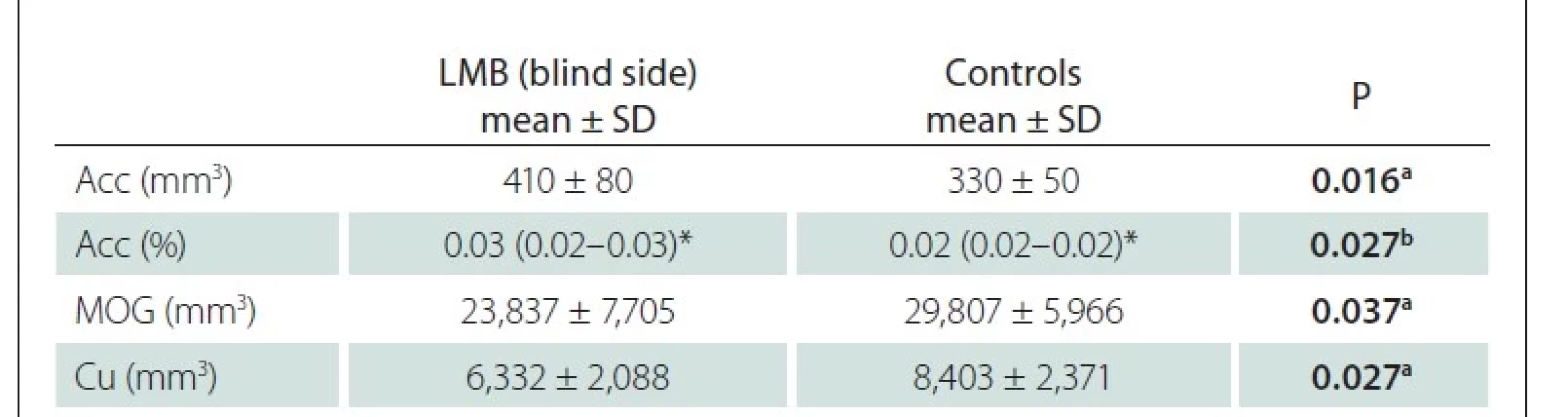
Acc – accumbens; Cu – cuneus; LMB – late-onset monocular blindness; MOG – middle occipital
gyrus; SD – standard deviation
In a comparison of cerebral volumes of the sighted eye side of the LMB group and ipsilateral cerebral volumes of controls, we found lower SMG volumes, higher Sb volumes in the LMB group than in the control group (Tab. 4). There was no significant difference in other parameters.

LMB – late-onset monocular blindness; Sb – subiculum; SD – standard deviation; SMG – supramarginal
gyrus
Discussion
MRICloud and volBrain are a web-based platform for automated brain segmentation and distributed remote computing. Automatic cerebral segmentation is an emerging technology which shows good accuracy when compared to gold standard segmentation [14]. VolBrain CERES is one of the new applications among automatic cerebral/cerebellar segmentation applications. Romero et al reported that volBrain CERES outperforms existing cerebrum/cerebellum segmentation methods such as SUIT, MAGeT and RASCAL, and produces accurate results [15].
In the patients with LMB who participated in our research, we discovered that the Cu and cornu ammonis-2 and cornu ammonis-3 (CA2, CA3) of the hippocampus had smaller sizes in the blind sides, compared with sighted sides. When seen from an anatomical standpoint, CA2, CA3, and the DG are believed to be a single region referred to as CA23DG. This region is the one that receives some inputs from the entorhinal cortex via the perforant path [20]. The other structure that was identified in our research is called the Cu, and it is situated on the medial surface of the cerebral hemisphere. It is responsible for receiving visual information from the superior quadrantic retina on the same side, which corresponds to the inferior VF on the other side. The Cu and LG are the anatomical locations of the primary visual cortex, also known as the Brodmann area 17 [21]. When taken in this light, the location of this volumetric loss may be connected with the adaptive changes that occur in patients who are monocularly blind.
From the ophthalmologic perspective, it has been demonstrated that there is no difference in the optic nerve, retina and choroid OCT findings in the sighted eyes of subjects with long-term monocular blindness [22]. However, early-onset, in particular from birth, binocular blind persons may experience several neuroanatomical and functional changes in the brain. To the best of our knowledge, previous studies have not differentiated the side of the blindness, as we have selected to do in our study, possibly because previous studies mostly included binocular vision loss, or congenital blindness. On the other hand, only a few studies, with very limited data, have investigated the alterations in late-onset, advanced monocular individuals [23,24]. In one of these cases, similar to our study, researchers investigated the cerebellar volumes between eleven patients with LMB and eleven healthy controls. They compared the cerebellar structures between the affected eye in the LMB group and similar sides in the control group. Mean cerebellar cortical thickness, cerebellar lobule VI-cerebellar lobule crus I-II cortical thickness and cerebellar lobule VI GM volume (GMV) values were found to be lower in the LMB group [23]. For our part, we compared both sides in the same patient and investigated all the cerebral structures, not solely the cerebellum.
The optic nerves, the lateral geniculate nucleus, the pericalcarine WM and the corpus callosum were identified as the brain localizations that decreased, in patients who had binocular blindness [25–30]. There is a volumetric decrease in the superior lateral occipital cortices, which are crucial to depth perception, in monocular individuals due to the trauma or tumor surgery, as indicated in a previous study conducted by Prins et al [12]. In their study, they used high-resolution T1-weighted MRI and voxel- and surface-based morphometry to compare GM and WM volume (WMV), cortical thickness, mean curvature and surface area, between fifteen monocularly blind patients and eighteen healthy controls. Their inclusion criteria were the same as ours, with at least 5 years of acquired monocular blindness. In our study, we included monocular blind patients who could see with the healthy side, in order to examine whether any changes in the sighted and blind sides’ cerebral volumes could be observed.
In another study, monocular blind individuals showed significantly decreased regional homogeneity (ReHo) values in the right rectal gyrus, right Cu, right anterior cingulate and the right lateral occipital cortex in an fMRI analysis, compared with sighted individuals. On the other hand, monocular blind individuals showed increased ReHo values in the right inferior temporal gyrus, right frontal middle orbital, left posterior cingulate/precuneus. The researchers concluded that LMB caused some functional abnormalities in the visual cortex and other vision-related brain regions, which may indicate that these regions of the brain are not functioning properly [31]. For their part, Li et al evaluated spontaneous brain activity in persons with LMB by employing resting-state fMRI and the amplitude of low-frequency fluctuation (ALFF) methods. Both techniques measure brain activity when the subject is at rest. They found lower ALFF values in the left cerebellum anterior lobe, right parahippocampal gyrus, right Cu, left precentral gyrus and left paracentral lobule in LMB individuals, compared with sighted individuals. Conversely, they found high ALFF values in the right/left middle frontal gyrus, and in the left SMG [32]. When we compared the blind sides of persons with LMB with the ipsilateral data of the healthy control groups, we discovered increased volumes of the Acc and its ratio to the cerebral cavity. On the other hand, we identified decreased volumes of MOG and Cu. In addition, we found lower volumes of the SMG and the higher volumes of the Sb in the LMB group, when compared to the cerebral volumes of the sighted side of the LMG group and the sizes of the controls’ ipsilateral cerebral volumes.
Because of its output and cytochemical similarity to the motor nuclei of the basal ganglia, the nucleus accumbens is considered to be the neural interface between the limbic and motor systems. This suggests that the nucleus accumbens is an important factor in the control of the biological drives necessary for survival, and indeed, studies have demonstrated that the Acc plays crucial roles in locomotion, as well as motivational and rewarding learning [33]. In a similar manner, the Sb, which is the principal output structure of the hippocampus, plays a significant role in both the processing of mnemonics and the navigation in space [34]. In this regard, our findings uncovered a function for the Acc and the Sb, both of which have well-established anatomical localizations, but are ignored in studies of persons who are monocularly blind. For example, an increase in the volume of the Sb may be considered a fundamental component of the superior spatial navigation in these individuals. For the purpose of confirming these anatomical alterations, it would be enticing to explore the functional and metabolic changes, as well as the cognitive qualities of monocular individuals. Although it is not exactly known whether there is a difference in visual anatomy and visual cortex between LMB and sighted individuals, we discovered reduced amounts of SMG, MOG in the LMB group in our study. According to the findings of Shi and colleagues, patients with monocular blindness have aberrant WMV and GMV, which can be seen as local alterations in the brain. In addition, decreased GMV in particular regions of the brain was found to be linked with the length of blindness [35]. This finding may point to neuropathological mechanisms that underlie the loss of vision experienced by individuals who have monocular blindness.
The current investigation offered useful structural information; nevertheless, the sample size was limited and thus a larger cohort may have produced more substantial results. Another limitation of our study was the wide range of blindness period (8 to 32 years), as the structural changes of the brain might develop during the time and therefore, such a wide range could represent a source of bias. A further disadvantage of this study was the absence of clinical data, such as neuropsychological tests, to measure skills such as memory, attention and visuospatial memory. It would be interesting to corroborate our findings with fMRI and clinical features.
Conclusion
Advanced monocular blindness may cause alterations in the cerebral structures, such as increasing volumes in the Sb and Acc, and decreased volumes in the SMG, MOG. To corroborate these structural alterations, it is necessary to analyze the functional metabolism of these areas.
Ethical principles
The entire study was conducted in accordance with the Helsinki Declaration of 1975 (as revised in 2004 and 2008). It has been approved by the Medical Ethics Committee of Alanya Alaaddin Keykubat University, Turkey with the research No. 10354421/2019/2 (February 7, 2019). Written informed consent has been obtained from all patients.
Conflict of interest
The authors declare they have no potential conflicts of interest concerning drugs, products, or services used in the study
Ece Özdemir Öktem, MD
Alanya Alaaddin Keykubat University
Department of Neurology
Antalya
Turkey
e-mail: ece.oktem@alanya.edu.tr
Accepted for review: 25. 10. 2022
Accepted for print: 2. 3. 2023
Sources
1. Mirza GD, Okka M, Mirza E et al. The causes and frequency of monocular and binocular blindness in adults applying to the health committee of a University Hospital in Central Anatolia. Turk J Ophthalmol 2021; 51 (5): 282–287. doi: 10.4274/tjo.galenos.2020.88120.
2. World Health Organization. World report on vision 2019. [online]. Available from: https: //www.who.int/publications/i/item/9789241516570.
3. World Health Organization. Global ınitiative for the elimination of avoidable blindness: action plan 2006–2011. [online]. Available from: http: //www.who.int/blindness/Vision2020 _report.pdf.
4. Pararajasegaram R. VISION 2020-the right to sight: from strategies to action. Am J Ophthalmol 1999; 128 (3): 359–360. doi: 10.1016/s0002-9394 (99) 00251-2.
5. Stingl K, Greppmaier U, Wilhelm B et al. Subretinal visual implants. Klin Monbl Augenheilkd 2010; 227 (12): 940–945. doi: 10.1055/s-0029-1245830.
6. MacLaren RE, Groppe M, Barnard AR et al. Retinal gene therapy in patients with choroideremia: initial findings from a phase 1/2 clinical trial. Lancet 2014; 383 (9923): 1129–1137. doi: 10.1016/S0140-6736 (13) 62117-0.
7. Carroll, TJ. Blindness: what it is, what it does, and how to live with it. Boston: Little; Brown and Company 1968.
8. Wagner-Lampl A, Oliver GW. Folklore of blindness. J Visual Impairment Blindness 1994; 88 (3): 267–276. doi: 10.1177/0145482X9408800312.
9. Merabet LB, Pascual-Leone A. Neural reorganization following sensory loss: the opportunity of change. Nat Rev Neurosci 2010; 11 (1): 44–52. doi: 10.1038/nrn2758.
10. Fortin M, Voss P, Lord C et al. Wayfinding in the blind: larger hippocampal volume and supranormal spatial navigation. Brain 2008; 131 (11): 2995–3005. doi: 10.1093/brain/awn250.
11. Stevens AA, Weaver KE. Functional characteristics of auditory cortex in the blind. Behav Brain Res 2009; 196 (1): 134–138. doi: 10.1016/j.bbr.2008.07.041.
12. Prins D, Jansonius NM, Cornelissen FW. Loss of binocular vision in monocularly blind patients causes selective degeneration of the superior lateral occipital cortices. Invest Ophthalmol Vis Sci 2017; 58 (2): 1304–1313. doi: 10.1167/iovs.16-20404.
13. Aguirre GK, Datta R, Benson NC et al. Patterns of individual variation in visual pathway structure and function in the sighted and blind. PLoS One 2016; 11 (11): e0164677. doi: 10.1371/journal.pone.0164677.
14. Avnioglu S, Velioglu HA, Cankaya S et al. Quantitative evaluation of brain volumes in drug-free major depressive disorder using MRI cloud method. Neuroreport 2021; 32 (12): 1027–1034. doi: 10.1097/WNR.0000000000001682.
15. Rezende TJR, Campos BM, Hsu J et al. Test-retest reproducibility of a multi-atlas automated segmentation tool on multimodality brain MRI. Brain Behavior 2019; 9 (10): e01363. doi: 10.1002/brb3.1363.
16. Manjón JV, Romero JE, Vivo-Hernando R et al. Vol2Brain: a new online pipeline for whole brain MRI analysis. Front Neuroinform 2022; 16: 862805. doi: 10.3389/fninf.2022.862805.
17. Coupé P, Manjón JV, Fonov V et al. Patch-based segmentation using expert priors: application to hippocampus and ventricle segmentation. Neuroimage 2011; 54 (2): 940–954. doi: 10.1016/j.neuroimage.2010.09.018.
18. Park MT, Pipitone J, Baer L et al. Derivation of high--resolution MRI atlases of the human cerebellum at 3T and segmentation using multiple automatically generated templates. Neuroimage 2014; 95: 217–231. doi: 10.1016/j.neuroimage.2014.03.037.
19. Romero JE, Coupé P, Giraud R et al. CERES: a new cerebellum lobule segmentation method. Neuroimage 2017; 147: 916–924. doi: 10.1016/j.neuroimage.2016.11.003.
20. Izci Y, Erbas YC. Hippocampus: its structure and functions. Turkish Neurosurg J 2015; 25 (3): 287–295.
21. Gövsa F. Morphology of the human cuneus by magnetic resonance imaging. Turkish J Med Sci 1998; 28 (4): 401–406.
22. Oktem C, Aslan F, Oktem EO. Evaluation of the effect of unilateral late blindness on the retina, optic nerve and choroid parameters in the sighted eye. Int Ophthalmol 2021; 41 (12): 4083–4089. doi: 10.1007/s10792-021-01981-0.
23. Hanson RLW, Gale RP, Gouws AD et al. Following the status of visual cortex over time in patients with macular degeneration reveals atrophy of visually deprived brain regions. Invest Ophthalmol Vis Sci 2019; 60 (15): 5045–5051. doi: 10.1167/iovs.18-25823.
24. Özen Ö, Aslan F. Morphometric evaluation of cerebellar structures in late monocular blindness. Int Ophthalmol 2021; 41 (3): 769–776. doi: 10.1007/s10792-020-01629-5.
25. Cecchetti L, Ricciardi E, Handjaras G et al. Congenital blindness affects diencephalic but not mesencephalic structures in the human brain. Brain Struct Funct 2016; 221 (3): 1465–1480. doi: 10.1007/s00429-014-0984-5.
26. Noppeney U, Friston KJ, Ashburner J et al. Early visual deprivation induces structural plasticity in grey and white matter. Curr Biol 2005; 15 (13): 488–490. doi: 10.1016/j.cub.2005.06.053.
27. Pan WJ, Wu G, Li CX et al. Progressive atrophy in the optic pathway and visual cortex of early blind Chinese adults: a voxel-based morphometry magnetic resonance imaging study. Neuroimage 2007; 37 (1): 212–220. doi: 10.1016/j.neuroimage.2007.05.014.
28. Ptito M, Schneider FC, Paulson OB et al. Alterations of the visual pathways in congenital blindness. Exp Brain Res 2008; 187 (1): 41–49. doi: 10.1007/s00221-008-1273-4.
29. Leporé N, Voss P, Lepore F et al. Brain structure changes visualized in early-and late-onset blind subjects. Neuroimage 2010; 49 (1): 134–140. doi: 10.1016/ j.neuroimage.2009.07.048.
30. Tomaiuolo F, Campana S, Collins DL et al. Morphometric changes of the corpus callosum in congenital blindness. PLoS One 2014; 9 (9): e107871. doi: 10.1371/journal.pone.0107871.
31. Huang X, Ye CL, Zhong YL et al. Altered regional homogeneity in patients with late monocular blindness: a resting-state functional MRI study. Neuroreport 2017; 28 (16): 1085–1091. doi: 10.1097/WNR.0000000000000855.
32. Li Q, Huang X, Ye L et al. Altered spontaneous brain activity pattern in patients with late monocular blindness in middle-age using amplitude of low-frequency fluctuation: a resting-state functional MRI study. Clin Interv Aging 2016; 11: 1773–1780. doi: 10.2147/CIA.S117292.
33. Salgado S, Kaplitt MG. The nucleus accumbens: a comprehensive review. Stereotact Funct Neurosurg 2015; 93 (2): 75–93. doi: 10.1159/000368279.
34. O‘Mara SM, Commins S, Anderson M et al. The subiculum: a review of form, physiology and function. Prog Neurobiol 2001; 64 (2): 129–155. doi: 10.1016/s0 301-0082 (00) 00054-x.
35. Shi WQ, He Y, Li QH et al. Central network changes in patients with advanced monocular blindness: a voxel--based morphometric study. Brain Behav 2019; 9 (10): e01421. doi: 10.1002/brb3.1421.
Labels
Paediatric neurology Neurosurgery NeurologyArticle was published in
Czech and Slovak Neurology and Neurosurgery

2023 Issue 2
Most read in this issue
- Current and future therapeutic options for the treatment of the generalized form of myasthenia gravis
- The problematics of post-stroke disability assessment
- Cenobamát v léčbě farmakorezistentní fokální epilepsie
- Standardizované a pokročilé techniky MR v diagnostice dětských nádorů mozku
